 Aquatic Insects of Michigan
Aquatic Insects of Michigan
by Ethan Bright, Museum of Zoology Insect Division and School of Natural Resources and Environment
University of Michigan
- Home
- Species Lists
- Coleoptera
- Diptera
- Ephemeroptera
- Acanthametropodidae
- Ameletidae
- Ametropodidae
- Arthropleidae
- Baetidae
- Baetiscidae
- Behningiidae
- Caenidae
- Ephemerellidae
- Ephemeridae
- Heptageniidae
- Isonychiidae
- Leptohyphidae
- Leptophlebiidae
- Metretopodidae
- Neoephemeridae
- Oligoneuriidae
- Palingeniidae
- Polymitarcyidae
- Potamanthidae
- Pseudironidae
- Siphlonuridae
- Heteroptera
- Hymenoptera
- Lepidoptera
- Megaloptera
- Neuroptera (Sisyridae)
- Odonata
- Orthoptera
- Plecoptera
- Trichoptera
- Other Orders
- Keys/Identification
- Aquatic Insects of Michigan
Neuroptera (Sisyridae - Spongillaflies) of Michigan - Identification
Introduction
Neuroptera is a very large, almost exclusively terrestrial order. This group contains two families with aquatic stages. Nevrorthidae contains three genera found in southern Europe, east Asia, and Australia, respectively. Only Sisyridae - the spongillaflies - are found in North America, and 3 species in 2 genera are found in Michigan.Larvae of Climacia areolaris are most common in among sponge colonies in streams and wave-swept margins of lakes; adults are small, net-veined, and brownish. The larvae of Sisyra (fuscata, vicara) occur in sponges in small ponds or still waters.
Only the the larval stage is adapted for aquatic living. Females lay eggs enmeshed with silk on vegetation overhanging an aquatic body (lentic or lotic). Upon hatching, the larvae drop into the water and search for freshwater sponges upon which they prey by using long, specialized stylet mouthparts to pierce and suck out contents. Chandler (1956:234-235)) has a nice synopsis of larval-pupal life history:
"Hatching occurs [within] about eight days. The larva escapes from the egg by means of an egg-burster situated on the tip of the amnioci membrane which it soon sheds. It then drops upon the surface of the water and, often with considerabgle difficulty, forces its way through the surface film. It may crawl on the under side of the surface film, swim by curling the abdomen ventrally and snapping it out straight, float or sink at will, or wander about on the bottom. The sponge host is found in one of these ways. Once settled upon a sponge, the larvae do not readily leave it unless the sponge dies. They frequently enter the ostioles of the sponge. The first larva commonly feeds for one-half to two minutes, rests or wanders about for one to five minutes, then reinserts its mouth parts for another meal. The sucking mouth parts consist of the mandibles and. maxillae on each side joined so as to make a tube. The right and left pairs can also be joined. Color varies from green to yellow-brown depending somewhat on the color of the sponge. The mature third larvae crawl or swim to an object upon which they can emerge from the water. Most larvae emerge from one to five hours after dark. They may travel as far as fifty feet before selecting a site to spin a cocoon, usually in a somewhat protected location. The larvae take a firm stance and do not move their feet while spinning the outer net. The tenth abdominal segment is modified into a spinneret with the silk exuding from the anus. The spinning is accomplished by bending the abdomen this way and that with some telescopic movement. The resulting hemispherical outer net has a radius about the same length as that of the abdomen. In Climacia this outer net has a delicate hexagonal mesh whereas in the fibers are merely crisscrossed. The cocoon is about half the diameter of the outer net and is composed of crisscrossed fibers in both genera. The cast pupal skin is wadded into the end of the cocoon. The sex of the pupa can be determined by examining the genitalia. The mandibles are heavily sclerotized and used by the pupa to chew its way out of the cocoon and net. The appendages are immovable until just before emergence, which usually occurs after sunset. Within two hours the insect assumes its adult form and color."
Larvae are very small (< 8 mm), so one most commonly finds them by searching for the sponges. There are 3 instars. With a small development range and rapid life cycle, multivoltism (more than one generation per year) is common with spongillaflies. Like Megaloptera, larvae crawl out of the aquatic environment to create a pupal chamber. Generally, sisyrids overwinter as larva, but pre-pupae - encased in their silken pupal cocoons - may overwinter to emerge next spring. Unlike Megaloptera, larvae generally travel longer distances and construct a silken cocoon in which to pupate. (During this stage in particular spongillaflies are susceptible to predation and parasitism - pteromalid wasps of Sisyridivora parasitize Climacia larvae and pupae in their cocoons (Pupedis 1978)). Because larvae lack a gut, the adult must defecate to empty its gut. Adults are short-lived (2-3 weeks), tend to be crepuscular or nocturnal, and feed on pollen or nectar until mating and oviposition.
Freshwater sponges, which are filter-feeders, are also directly preyed upon by other aquatic insects, in particular species of the caddisfly genus Ceraclea (Leptoceridae) and genera of chironomid midge larvae, in particular Xenochironomus xenolabis.
Adults (adapted from Parfin and Gurney 1956, Evans and Neunzig 1996)
| 1a | a. Rs of forewing with one fork before the pterostigmata | Climacia areolaris (Hagen) |
| b. Forewing with brown markings | ||
| c. Last segment of the labial palp similar in size and shape as the other segments | ||
| 1b | a. Rs of forewing with more than one fork before the pterostigmata | Sisyra McLachlan, 2 |
| b. Forewing uniformily brown | ||
| c. Last segment of the labial palp greatly enlarged, triangular in shape | ||
| 2a(1b) | a. Male claspers long, wide at base, then sharply curved and gradually narrowed to a terminal point, with the basal plate about 1/3 as long as the claspers (Fig. 1) | Sisyra fuscata (Fabricius) |
| b. Lateroventral halves of Ab9 tergite of female more or less triangular, rounded ventrally and with one prominent, posterior, sclerotized articulation ridge (Fig. 2) | ||
| c. Forewing of both sexes about 5mm long and 2 mm wide, usually with the R2 not deeply forked (i.e., basal or level to the junction point of Sc and R1), apex oval, symmetrical with respect to the longitudinal axis of the wing | ||
| d. Vertex, frons, antennae and thorax blackish | ||
| e. Basal antennal segment with a sharp inner curve | ||
Fig. 1 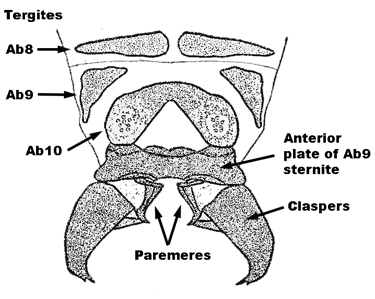 Fig. 2 Fig. 2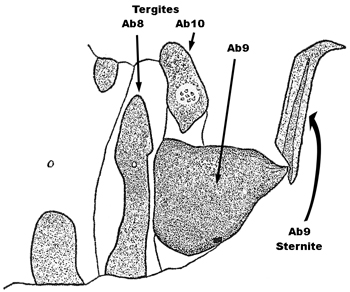 |
||
| 2b | a. In dorsal view, male claspers short, squat, with the basal plate about as long as the claspers (Fig. 3) | Sisyra vicaria (Walker) |
| b. Lateroventral halves of Ab9 tergite of female elongate, with both anterior and posterior sclerotized articulation ridges (Fig. 4) | ||
| c. Forewing of both sexes often > 5mm long and > 2 mm wide, usually with the R2 usually forked (i.e., basal to the junction point of Sc and R1), apex more acute, and less symmetrical with respect to the longitudinal axis of the wing than above | ||
| d. Vertex, frons, and thorax yellow to yellow-brown, with the vertex having a thin dark line encircling the basal antennal segments, and teh basal segments of the antennae dark brown, with the remaining segments yellow or brown to the midpoint and then yellowish to the apex | ||
| e. Basal antennal segment smoothly curved on the inner margin | ||
Fig. 3 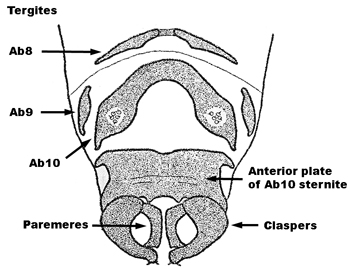 Fig. 4 Fig. 4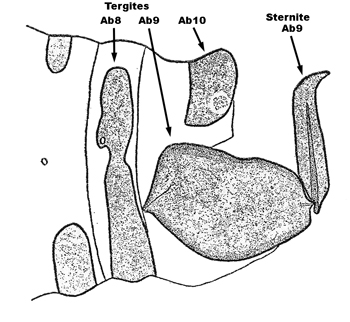 |
||
Pupae
(adapted from Evans and Neunzig 1996)
| 1a | Segments of labial palp similar | Climacia areolaris (Hagen) |
| 1b | Last palpal segment of labium greatly enlarged and triangular | Sisyra McLachlan |
Larvae (adapted from Cuyler 1958, Evans and Neunzig 1996, Neunzig 1966)
| 1a | a. Pair of dorsal setae present on Ab8 | Climacia areolaris (Hagen) |
| b. Ventral pair of medial setae on Ab8 raised on tubercles, and only slightly closer together than those on Ab9 | ||
| c. Small acute spines at the bases of thoracic setae | ||
| 1b | a. Pair of dorsal setae absent on Ab8 | Sisyra McLachlan |
| b. Ventral pair of medial setae on Ab8 sessile, and distinctly closer together than those on Ab9 | ||
| c. Small acute spines at the bases of thoracic setae absent | ||
Diagnoses
(From Parfin and Gurney 1956)
Head with vertex yellow, occasionally with some yellowish brown, sometimes a longitudinal, dark brown streak immediately behind and adjacent to pronotal margin (not always visible on pinned specimens); face yellow, sometimes light brownish; palpi yellow; antennae blackish brown, often becoming slightly lighter at the apices (approximately 57-62 segments) ;legs yellow, with mesothoracic and metathoracic coxae brown; pronotum yellowish to medium brown dorsally, sometimes with central blackish brown streaking,
mesothoracic and metathoracic pleura darker brown; abdomen brownish. Forewing:  Average length of female 5.2 mm, of male 4.6; average width of female 1.8 mm, of male 1.48 mm; membrane yellowish with two prominent and two less conspicuous blackish brown spots and three prominent yellowish to hyaline areas; the first large dark brown patch, the basal radiomedial streak, longitudinally elongate and usually beginning in the subcostal space about where Sc and R almost meet, continuing along to R+MA or almost to Rs posteriorly, extending laterally to vicinity of 1st r, the second an oblique antepterostigmal spot directed basally, usually broadest at the costa, then proceeding down along and narrowing at 2d r and 1st r-m with distal portion of subcostal area usually dark brown but sometimes almost clear, the two spots more or less interconnected by intervenational streaks from R1 to MP: the third dark brown spot sometimes indistinct, a small postpterostigmal spot, and the fourth, a small basal anal patch along inner margin; longitudinal intervenational brownish streaks along apical fourth and entire outer and inner margins of wing from pterostigma down except sometimes clear near Cu1 and Cu2; all veins brown in general except for portions in the yellowish areas of the wing and middle thirds of R2 and R3; pterostigma with long central yellowish area covering seven or eight costal cross-veins between and antepterostigmal postpterostigmal spots; Rs usually forking into R2+3 and R4+5 below pterostigma slightly distad of or at about level of point of coalescence of Sc and R1; MA with terminal fork below pterostigma; MP1+2 usually forking proximal to or at about level of point of coalescence of Scand R1, and forking into uneven fork, with first fork closer to stem and with first branch of first fork at an angle; Cu1 with four to six branches to margin (five most common); about 12 or 13 costal crossveins before pterostigma; three radial cross-veins, 1st r usually basad of midpoint of free stem of Rs, 2d r to Rs, 3d r below pterostigma to Rs, R2+3 or to fork; 2d r-m usually slightly distad of 3d r (occasionally directly under) and Rs fork, the outer gradates (five) being irregular at this point; 2d m between MA and MP1+2 usually slightly distad of, occasionally directly under, 1st r-m; 3d m long, obliquely directed basad so as to make cell between 2d and 3d m as an inverted trapezoid; the inner gradates also in irregular series. Hindwing: Average length of female 4 mm, male 3.8 mm, width of female 1.5 mm, male 1.4 mm; membrane hyaline to yellowish; two prominent dark brown spots, the first larger (antepterostigmal spot) and the second smaller (postpterostigmal spot); faint brownish shading along about first four costal cross-veins; center of pterostigma with long yellowish portion covering seven to eight costal cross-veins; brownish veins include radial veins beyond basal forking of R, MA, distal portions of MP, cubital and anal veins (the basal portions of these veins and 1st m usually clear), the radial and anal cross-veins, outer gradates, and basal sinuous piece of MA; Rs usually forking into R2+3 and R4+5 at point basad of point of coalescence of Sc and R1; Cu1 with three to five branches reaching margin; six outer gradates, including a sectoral cross-vein. Male genitalia: Eighth tergite longer dorsally, tapering lateroventrally, not as broad as ninth tergite; ninth tergite shorter mediodorsally and about as long ventrally and as wide as tenth tergite; tenth tergite with mediodorsal border of each half short, the ventrolateral border widened and prolonged distally; ninth sternite with appearance of a pair of median laterally curved processes extending distally from inner surface; two halves of tenth sternite almost striplike, then turning inwardly and broadened ventrally, covered with coarse, toothlike, setae-bearing papillae; parameres with a more or less diamond-shaped flap projecting usually anterodorsally from median plate, anterior margin heart shaped, posterior hooks tripartite, largest process toward exterior, smallest medial. Female genitalia: Tergite longer dorsally and shorter ventrally, anterior margin more or less convex; ninth tergite with each lateroventral half large, moderately elongate, anterior margin broadest with slight indentation near dorsal border, dorsal border gradually sloped from proximal to distal border, ventral margin rounded, with a slight irregularity prior to articulation point.
Average length of female 5.2 mm, of male 4.6; average width of female 1.8 mm, of male 1.48 mm; membrane yellowish with two prominent and two less conspicuous blackish brown spots and three prominent yellowish to hyaline areas; the first large dark brown patch, the basal radiomedial streak, longitudinally elongate and usually beginning in the subcostal space about where Sc and R almost meet, continuing along to R+MA or almost to Rs posteriorly, extending laterally to vicinity of 1st r, the second an oblique antepterostigmal spot directed basally, usually broadest at the costa, then proceeding down along and narrowing at 2d r and 1st r-m with distal portion of subcostal area usually dark brown but sometimes almost clear, the two spots more or less interconnected by intervenational streaks from R1 to MP: the third dark brown spot sometimes indistinct, a small postpterostigmal spot, and the fourth, a small basal anal patch along inner margin; longitudinal intervenational brownish streaks along apical fourth and entire outer and inner margins of wing from pterostigma down except sometimes clear near Cu1 and Cu2; all veins brown in general except for portions in the yellowish areas of the wing and middle thirds of R2 and R3; pterostigma with long central yellowish area covering seven or eight costal cross-veins between and antepterostigmal postpterostigmal spots; Rs usually forking into R2+3 and R4+5 below pterostigma slightly distad of or at about level of point of coalescence of Sc and R1; MA with terminal fork below pterostigma; MP1+2 usually forking proximal to or at about level of point of coalescence of Scand R1, and forking into uneven fork, with first fork closer to stem and with first branch of first fork at an angle; Cu1 with four to six branches to margin (five most common); about 12 or 13 costal crossveins before pterostigma; three radial cross-veins, 1st r usually basad of midpoint of free stem of Rs, 2d r to Rs, 3d r below pterostigma to Rs, R2+3 or to fork; 2d r-m usually slightly distad of 3d r (occasionally directly under) and Rs fork, the outer gradates (five) being irregular at this point; 2d m between MA and MP1+2 usually slightly distad of, occasionally directly under, 1st r-m; 3d m long, obliquely directed basad so as to make cell between 2d and 3d m as an inverted trapezoid; the inner gradates also in irregular series. Hindwing: Average length of female 4 mm, male 3.8 mm, width of female 1.5 mm, male 1.4 mm; membrane hyaline to yellowish; two prominent dark brown spots, the first larger (antepterostigmal spot) and the second smaller (postpterostigmal spot); faint brownish shading along about first four costal cross-veins; center of pterostigma with long yellowish portion covering seven to eight costal cross-veins; brownish veins include radial veins beyond basal forking of R, MA, distal portions of MP, cubital and anal veins (the basal portions of these veins and 1st m usually clear), the radial and anal cross-veins, outer gradates, and basal sinuous piece of MA; Rs usually forking into R2+3 and R4+5 at point basad of point of coalescence of Sc and R1; Cu1 with three to five branches reaching margin; six outer gradates, including a sectoral cross-vein. Male genitalia: Eighth tergite longer dorsally, tapering lateroventrally, not as broad as ninth tergite; ninth tergite shorter mediodorsally and about as long ventrally and as wide as tenth tergite; tenth tergite with mediodorsal border of each half short, the ventrolateral border widened and prolonged distally; ninth sternite with appearance of a pair of median laterally curved processes extending distally from inner surface; two halves of tenth sternite almost striplike, then turning inwardly and broadened ventrally, covered with coarse, toothlike, setae-bearing papillae; parameres with a more or less diamond-shaped flap projecting usually anterodorsally from median plate, anterior margin heart shaped, posterior hooks tripartite, largest process toward exterior, smallest medial. Female genitalia: Tergite longer dorsally and shorter ventrally, anterior margin more or less convex; ninth tergite with each lateroventral half large, moderately elongate, anterior margin broadest with slight indentation near dorsal border, dorsal border gradually sloped from proximal to distal border, ventral margin rounded, with a slight irregularity prior to articulation point.
Head with vertex blackish brown, shining, narrow median blackish streak over coronal sulcus usually not pronounced; face blackish brown with the exception of the clypeus and labrum, which are yellowish; palpi brownish; antennal segments brownish black to black, of approximately 46 segments; legs yellowish to light brownish with mesothoracic and metathoracic coxae brown; thorax blackish brown; abdomen blackish brown. 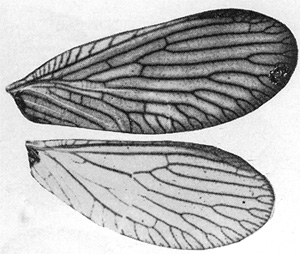 Forewing: Length female forewing 5 mm, of male 4.7 mm; average width of female forewing 2.1 mm, male 2 mm; apex rounded, oval; membrane more or less uniformly light brown (sometimes faint indications of light brown streaks, particularly near cubital areatoward inner margin) longitudinal veins brown; pterostigma slightly darker than membrane; R2 usually not forked basad to level of junction of Sc and R1; R4+5 usually forked more deeply from margin than R3, distad to or at about level of junction of Sc and R1; MP1+2 usually forking into almost even branches to margin; Cu1 with 3-5 branches to margin; approximately 11-15 costal cross-veins before pterostigma; usually three radial cross-veins; 1st r above and near midpoint of Rs (occasionally near fork of R4+5 from Rs), 2d r usually above R2 near forking of R2 and R3 before fork (occasionally to Rs) and 3rd r to R2 or R2 terminal fork, below pterostigma; marginal forking well-developed. Hindwing: Length of female hindwing approximately 4.3 mm, of male 4.2 mm; average width of female hindwing 2 mm, of male 1.8 mm; membrane almost hyaline, tinted with light brown, pterostigma darker brown; R2, R3, and R4+5 with terminal forks usually distad of level of junction of Sc and R1; R2 and R4+5 usually forked more deeply than R3; MA forked basad of above point; Cu1 with about 4-6 branches to margin; usually two radial cross-veins, 1st r slightly distad of forking of R2 and R3 from Rs; 2d to R2 or R2 fork under near margin; marginal forking well-developed. Male genitalia: Eighth tergite appearing divided in two with almost invisible median portion; two parts of ninth tergite small, irregularly shaped, usually with a narrow posteroventral projection; tenth tergite with irregular lateroventral margins; ninth sternite large, long, heavily setosem, almost rectangular; tenth sternite with short basal plate, less than half as long as claspers; claspers moderately long, thick at base, with rounded curve preceding terminal portion which tapers almost to a point, two distal toothlike projections at end of each clasper; parameres "leglike," broad anterodorsally, then narrowing, becoming more twisted and diverging into terminal "foot" with "toes" pointing medially toward bases of claspers. Female genitalia: Eighth tergite short dorsally, lengthening above and shortening around spiracle and then gradually lengthening ventrally; ninth tergite with each lateroventral half large, more or less triangle shaped with one posterior articulation ridge, greatest width at proximal border, dorsal border sloping obliquely posteroventrally, ventral margin distinctly rounded; tenth tergite tapering strongly lateroventrally.
Forewing: Length female forewing 5 mm, of male 4.7 mm; average width of female forewing 2.1 mm, male 2 mm; apex rounded, oval; membrane more or less uniformly light brown (sometimes faint indications of light brown streaks, particularly near cubital areatoward inner margin) longitudinal veins brown; pterostigma slightly darker than membrane; R2 usually not forked basad to level of junction of Sc and R1; R4+5 usually forked more deeply from margin than R3, distad to or at about level of junction of Sc and R1; MP1+2 usually forking into almost even branches to margin; Cu1 with 3-5 branches to margin; approximately 11-15 costal cross-veins before pterostigma; usually three radial cross-veins; 1st r above and near midpoint of Rs (occasionally near fork of R4+5 from Rs), 2d r usually above R2 near forking of R2 and R3 before fork (occasionally to Rs) and 3rd r to R2 or R2 terminal fork, below pterostigma; marginal forking well-developed. Hindwing: Length of female hindwing approximately 4.3 mm, of male 4.2 mm; average width of female hindwing 2 mm, of male 1.8 mm; membrane almost hyaline, tinted with light brown, pterostigma darker brown; R2, R3, and R4+5 with terminal forks usually distad of level of junction of Sc and R1; R2 and R4+5 usually forked more deeply than R3; MA forked basad of above point; Cu1 with about 4-6 branches to margin; usually two radial cross-veins, 1st r slightly distad of forking of R2 and R3 from Rs; 2d to R2 or R2 fork under near margin; marginal forking well-developed. Male genitalia: Eighth tergite appearing divided in two with almost invisible median portion; two parts of ninth tergite small, irregularly shaped, usually with a narrow posteroventral projection; tenth tergite with irregular lateroventral margins; ninth sternite large, long, heavily setosem, almost rectangular; tenth sternite with short basal plate, less than half as long as claspers; claspers moderately long, thick at base, with rounded curve preceding terminal portion which tapers almost to a point, two distal toothlike projections at end of each clasper; parameres "leglike," broad anterodorsally, then narrowing, becoming more twisted and diverging into terminal "foot" with "toes" pointing medially toward bases of claspers. Female genitalia: Eighth tergite short dorsally, lengthening above and shortening around spiracle and then gradually lengthening ventrally; ninth tergite with each lateroventral half large, more or less triangle shaped with one posterior articulation ridge, greatest width at proximal border, dorsal border sloping obliquely posteroventrally, ventral margin distinctly rounded; tenth tergite tapering strongly lateroventrally.
Head with vertex usually yellow, occasionally some brown, narrow median blackish streak over coronal sulcus from posterior margin to midvertex usually distinct, usually a narrow blackish ring around antennal sockets; face yellowish; palpi yellowish to brownish; basal two antennal segments usually brown, remaining segments yellowish to end or brown for 18 segments (approximately) and then yellowish to end (approximately 42-50 segments altogether); legs yellow with coxae brown and sometimes femora and tibiae with some brown; notum medium brown, pleura brown; abdomen brown. 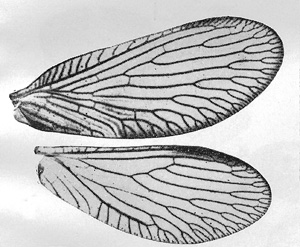 Forewing: Average length of female wing 5.5 mm, of male 5.4 mm, average width of female wing 2.4 mm, of male 2.3 mm; apex slightly asymmetrical with respect to longitudinal axis of wing; membrane more or less uniformly light brown (with occasionally a few indications of faint light brown streaks particularly near cubital and mediocubital areas toward inner margin); longitudinal and cross-veins brown; pterostigma slightly darker than membrane; R2 forked basad of or at level of junction of Sc and R1; R3 and R4+5 usually not forked as deeply from margin, although R4+5 sometimes with terminal fork near level of junction of Sc and R1; MP1+2 usually forking into two even branches to margin; Cu1 with three to five branches to margin; approximately 12-15 costal cross-veins before pterostigma; usually three radial cross-veins, 1st r above and usually near midpoint of free stem of R2, 2d r above R2 near forking of R2 and R3, and 3rd r below pterostigma to R2 or R2 terminal fork (in rare cases where fork is lacking); sometimes a 2d or even 3d (rarely) r-m present; marginal forking well-developed. Hindwing: Average length of female hindwing approximately 5 mm, male 4.6 mm, width female approximately 2.2 mm, of male 2 mm; membrane almost hyaline with pterostigma slightly darker brown than membrane; venation brown; R2, R3, and R4+4 usually with terminal forks distad of level of point of coalescence of Sc and R1; about three to seven branches of Cu1 to margin; two radial cross-veins, 1st r slightly distad of forking of R2 and R3 from Rs; 2d r to R2 or R2 fork under pterostigma near margin; marginal well-developed. Male genitalia: Eighth tergite with central portion short; two parts of ninth tergite small, more or less triangle shaped; tenth sternite with basal plate almost as long as claspers; claspers short, squat, curved, with sharp angular drop to terminal conical narrow portion, which is about one-third as wide as broad basal part of clasper, two setalike projections at end of each clasper; parameres "boot shaped." Female genitalia: Eighth tergite longer just above spiracle, shorter just below spiracle, then lengthening ventrally; ninth tergite with each lateroventral half large, longitudinally elongate, with one anterior and articulation ridge, dorsoproximal margin shaped like bird's head, dorsal border gradually sloped from proximal to distal border, ventral margin rounded.
Forewing: Average length of female wing 5.5 mm, of male 5.4 mm, average width of female wing 2.4 mm, of male 2.3 mm; apex slightly asymmetrical with respect to longitudinal axis of wing; membrane more or less uniformly light brown (with occasionally a few indications of faint light brown streaks particularly near cubital and mediocubital areas toward inner margin); longitudinal and cross-veins brown; pterostigma slightly darker than membrane; R2 forked basad of or at level of junction of Sc and R1; R3 and R4+5 usually not forked as deeply from margin, although R4+5 sometimes with terminal fork near level of junction of Sc and R1; MP1+2 usually forking into two even branches to margin; Cu1 with three to five branches to margin; approximately 12-15 costal cross-veins before pterostigma; usually three radial cross-veins, 1st r above and usually near midpoint of free stem of R2, 2d r above R2 near forking of R2 and R3, and 3rd r below pterostigma to R2 or R2 terminal fork (in rare cases where fork is lacking); sometimes a 2d or even 3d (rarely) r-m present; marginal forking well-developed. Hindwing: Average length of female hindwing approximately 5 mm, male 4.6 mm, width female approximately 2.2 mm, of male 2 mm; membrane almost hyaline with pterostigma slightly darker brown than membrane; venation brown; R2, R3, and R4+4 usually with terminal forks distad of level of point of coalescence of Sc and R1; about three to seven branches of Cu1 to margin; two radial cross-veins, 1st r slightly distad of forking of R2 and R3 from Rs; 2d r to R2 or R2 fork under pterostigma near margin; marginal well-developed. Male genitalia: Eighth tergite with central portion short; two parts of ninth tergite small, more or less triangle shaped; tenth sternite with basal plate almost as long as claspers; claspers short, squat, curved, with sharp angular drop to terminal conical narrow portion, which is about one-third as wide as broad basal part of clasper, two setalike projections at end of each clasper; parameres "boot shaped." Female genitalia: Eighth tergite longer just above spiracle, shorter just below spiracle, then lengthening ventrally; ninth tergite with each lateroventral half large, longitudinally elongate, with one anterior and articulation ridge, dorsoproximal margin shaped like bird's head, dorsal border gradually sloped from proximal to distal border, ventral margin rounded.
References
-
Chandler JP. 1956. Aquatic Neuroptera, pp. 234-236 in Usinger RL. Aquatic Insects of California, with keys to North American genera and California species. University of California Press, Berkeley, California, USA.
Evans ED, Neunzig HH. 1996. Megaloptera and Aquatic Neuroptera, pp. 298-308 in Merritt RW, Cummins KW (editors), An Introduction to the Aquatic Insects of North America, 3rd Edition. Kendall/Hunt Publishing Company: Dubuque, Iowa. 862 + xiii p.
Parfin SI, Gurney AB. 1956. The spongilla-flies, with special reference to those of the Western Hemisphere (Sisyridae, Neuroptera). Proceedings of the United States National Museum 105:421-529.
Pupedis RJ. 1978. Tube feeding by Sisyridivora cavigena (Hymenoptera: Pteromalidae) on Climacia areolaris (Neuroptera: Sisyridae). Annals of the Entomological Society of America 71(5):773-775.
Page created: April 14, 2006; Last edited: November 10, 2013 (EB)